Plants have specialized molecular mechanisms that allow them to recognize and respond to herbivory by releasing volatile plant hormones that attract predators and stunt the growth of the feeding insects. The discovery of this mechanism by Dr. Eric Schmelz has opened doors for the future of crop protection, given that the use of Bacillus thuringiensis transgenes is quickly becoming less effective.
Unlike mammals, plants cannot run away when threatened by a feeding pest. Although they cannot move, plants interact more with their ecosystems than you might expect. While humans protect their livelihood with warfare, one way plants protect themselves from being eaten is by releasing volatile compounds into their surroundings to attract defenders, such as parasitoids and predatory insects, to attack the damaging herbivores. Without a nose to smell or eyes to see, plants need specialized perception systems to specifically recognize when an insect takes a bite.
Over the course of human civilization, we have gone to great lengths to improve crop production; however, many modern year-round crops remain susceptible to pest attack. One method developed to protect plants from generalist pests is the introduction of bacterial Bacillus thuringiensis (Bt) transgenes that encode an insecticidal crystal (Cry) protein. There are many variations of Bt genes that encode diverse Cry toxins, each producing a variant toxin that targets different insects. Though this approach has successfully protected crops for some time, wide scale use of Bt genes created intense selective pressure that drives generalist insects to evolve adaptations to evade Cry toxin poisoning. As a result, our use of Cry toxins is at risk of becoming obsolete, rendering many crops vulnerable to insect attack. To combat pest evolution, researchers ventured to investigate whether naturally occurring plant defense mechanisms can be better leveraged to combat crop pests. A better understanding of dynamic plant defenses may lead to novel ways of effectively protecting crops in the future with diverse plant treatments.
Response to Attack
In 1990, scientists noticed that both corn and bean plants release distinct volatile odors minutes to hours after an insect takes a bite from them. Subsequent studies revealed that nearly all plants respond to herbivory by synthesizing and emitting volatile biochemicals, yet how insect attack was perceived remained unclear. Dr. Eric Schmelz, Professor of Cell and Developmental Biology at UC San Diego, in conjunction with Dr. Adam Steinbrenner and Dr. Alisa Huffaker, have recently identified a legume molecular recognition mechanism for insect herbivory.³
This molecular recognition mechanism is mediated by a particular plant receptor that binds to a specific plant protein fragment elicitor.³ Elicitors are compounds commonly found in caterpillar oral secretions deposited on the plant leaves during leaf digestion. When the plant receptor is activated by such an elicitor, this receptor stimulates the plant to synthesize and secrete many plant defense compounds. In bean plants, precisely defined oral secretion elicitors trigger plant volatile emissions. These emissions attract parasitoid wasps that use attacking caterpillars as hosts for their developing young. Therefore, through the oral secretion-stimulated production of volatile defense biochemicals, plants signal that their leaves are being chewed on and recruit natural enemy bodyguards that eliminate diverse groups of generalist pests.
The ability to precisely identify elicitor molecules that activate plant signaling cascades was crucial to understanding plant perception of insect herbivores. Dr. Schmelz’s contributions toward understanding the molecular cascade started in 2006 when he reported that cowpea plants recognize protein fragment elicitors derived from ATP synthase in fall armyworm oral secretions and initiate the rapid production of defense-related phytohormones, or plant hormones. The biochemical elicitors present in attacking herbivores, and perceived by plants, are the products of an ancient yet ongoing evolutionary arms race. The contents of insect oral secretions depend on their diet, yet bean plants have evolved adaptations to perceive highly conserved elicitor fragments from insects that vary very little across diverse plant groups.²
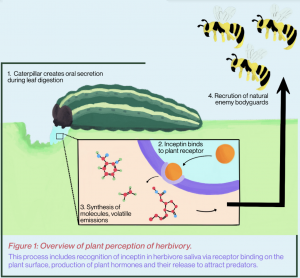
The cowpea plant’s response to herbivory and the content of herbivore secretions were characterized by capturing the released volatile biochemicals in a solvent, and upon heating and volatizing that solvent, deciphering the identities of the trapped biochemicals within. Other separation assays of insect oral secretion samples were used to identify the elicitors capable of triggering plant volatile hormone production. The Schmelz lab isolated the protein fragment inceptin from insect oral secretions, a protein derived during the decomposition of plant chloroplastic ATP synthase protein subunits. Inceptin contains a highly conserved amino acid motif present in nearly all green plant tissues. Thus, when armyworms chew on such green leaves, trace amounts of inceptin from the gut of the herbivore are released onto the wound surface, which activates a receptor that subsequently stimulates the synthesis of phytohormones like ethylene, jasmonate and salicylic acid. Each class of phytohormones then activates transcription factors that initiate the transcription and translation of various enzymes. These enzymes not only produce volatile biochemicals that attract predators but also anti-nutrients that reduce caterpillar growth rates.
Adaptation Arms Race
Over the years, several other elicitors were identified, which hints to a potentially vast diversity among plants’ pest-perception mechanisms. On one side of the battlefield, plants are evolving to recognize diverse compounds present in pest oral secretions, as evidenced by plant species recognizing molecules specific to arthropods. This recognition is derived from millions of years of plant-herbivore interactions driving the evolution of plant resistance.
However, herbivores are similarly engaged in their side of the evolutionary arms race. Specifically, herbivore evolution is driven by the significant selective pressure to create slight chemical changes in the oral secretion composition, enabling the pest to evade plant perception. Tracking this prediction, Dr. Schmelz identified a truncated and inactive form of inceptin that dominates in the oral secretions of the legume-specializing pest, the velvetbean caterpillar.¹ The altered inceptin elicitor containing a single amino acid loss still binds to the receptor yet fails to activate plant defense responses. Similar to a camouflaged attacker who uses elements of their environment to hide in plain sight, velvetbean caterpillars hide from bean plants by evading oral secretion recognition with altered inceptin elicitors. Thus, while plants are evolving methods to identify diverse pest oral secretions, pests themselves are evolving methods to hide from plant defense systems. This finding defined the role of molecular elicitors in triggering plant volatile emissions as adaptations evolved in the specialist pest velvetbean caterpillar to escape plants’ inceptin perception mechanisms. However, the underlying receptors on the plants involved in eliciting the defense mechanism itself remained unclear.
The Mysterious Receptor
By 2015, Dr. Schmelz and many other scientists were developing refined models of how herbivore-associated elicitors in oral secretions activate plant defense outputs. However, these models lacked the precise identity of the plant cell-surface receptor that binds inceptin. To identify this receptor, Dr. Schmelz and his team devised an assay to screen diverse bean germ cells for detectable phytohormone release in response to an inactive form of inceptin. This assay aimed to address whether wild plants are regaining ground in the evolutionary arms race against evolving pest oral secretions resistant to plant detection. The assay consisted of treating leaves with either water, inceptin, or inactive inceptin, and measuring defense responses to each. It was hypothesized that inactive inceptin would only elicit a plant defense response if the plant acquired mutations to allow it to detect this modified inceptin variant. Small yet reproducible plant responses were detected following application of the altered inceptin, suggesting that variation in the receptor gene could evoke variation in the plant elicitor’s perception.
Answers in Legume Genomes
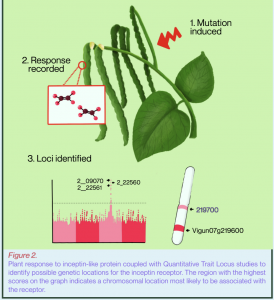 Once the model of herbivore perception by plants was validated, the next step was to locate the specific genetic locations, or loci, on the bean chromosomes that might contain inceptin receptor (INR) candidates. Following the careful analysis of 400 different bean plant lines and over 2000 biochemical samples, Dr. Schmelz and his group used both a Genome Wide Association Study (GWAS) and a separate Quantitative Trait Locus (QTL) mapping study to identify chromosome positions that were statistically associated with the rare positive plant elicitation responses to the inactive inceptin variant.² QTL mapping is a statistical description of the association of a chromosomal region with the phenotypic trait data, while GWAS involves looking at several plants of the same species and deciding which traits may be associated by genetic variation. Both mapping approaches identified the same shared genetic position associated with antagonist responsiveness located inside the coding region of a receptor-like protein candidate, termed the inceptin receptor (INR).
Once the model of herbivore perception by plants was validated, the next step was to locate the specific genetic locations, or loci, on the bean chromosomes that might contain inceptin receptor (INR) candidates. Following the careful analysis of 400 different bean plant lines and over 2000 biochemical samples, Dr. Schmelz and his group used both a Genome Wide Association Study (GWAS) and a separate Quantitative Trait Locus (QTL) mapping study to identify chromosome positions that were statistically associated with the rare positive plant elicitation responses to the inactive inceptin variant.² QTL mapping is a statistical description of the association of a chromosomal region with the phenotypic trait data, while GWAS involves looking at several plants of the same species and deciding which traits may be associated by genetic variation. Both mapping approaches identified the same shared genetic position associated with antagonist responsiveness located inside the coding region of a receptor-like protein candidate, termed the inceptin receptor (INR).
To understand whether the INR candidate interacts only with herbivory-associated molecules, an experiment was designed to compare INR’s mediated response to inceptin versus INR’s mediated response to a bacterial derived pathogen elicitor. Transgenic expression of INR in non-native tobacco plants demonstrated that plants transiently expressing INR proteins respond to inceptin, while bacterial elicitors did not activate any INR response, showing how such bacterial elicitors require different receptors. Therefore, newly imparted defense phytohormone release was only detected following inceptin treatment and predictable interactions with the INR.
Having identified the INR, the Schmelz group next investigated whether the INR gene is unique to cowpea and bean plants, and whether there are variants of the receptor conferring the same defense response in other species.² To determine this, they looked at shared genetic loci of different related legume plants with additional conserved marker genes nearby. It was found that only legumes with predicted INR genes responded to inceptin elicitation, while when expressed in tobacco, only candidate INR transgenes with more than 90% amino acid similarity produced defense responses. Closely related key crops such as soybean lack predicted functional copies of INR, and subsequently lack inceptin responses. Why soybean plants lost this perception mechanism remains a mystery.
Given that INR expression equipped plants with the ability to recognize attack, Dr. Schmelz examined the ability of distantly-related plants to acquire inceptin perception. Stable transgenic tobacco plants expressing INR demonstrated that the INR transgene could be stably transferred to distantly related plants that lack inceptin responses and successfully boosts the production of antiherbivore defenses following INR activation. This discovery shows promise for enhancing natural crop protection mechanisms by staking the best of diverse plant immunity traits.
Plant-driven Defense in Agriculture
Recent efforts in bean and corn plant models by Dr. Schmelz and Dr. Huffaker are pushing the boundaries of natural plant defense systems. Plants are incapable of fleeing from threats, yet their ability to call for help and bolster their defenses when they perceive danger extends their physical reach in a way that is not reproducible by humans. This defense behavior in plants has the potential to change the way we manage and optimize agricultural settings. The expression of diverse plant derived INR transgenes in naive crops enables us to boost natural protective responses of plants without further human inputs. The ability of a plant to perceive danger changes the interaction–an army that is not expecting attack is vulnerable. Receptors for common insect elicitors that activate plant defense responses provide a powerful radar, and are almost certain to be useful in bolstering crop protection in an environmentally sustainable fashion.
References
- Eric A. Schmelz, Alisa Huffaker, Mark J. Carroll, Hans T. Alborn, Jared G. Ali, Peter E.A. Teal, An Amino Acid Substitution Inhibits Specialist Herbivore Production of an Antagonist Effector and Recovers Insect-Induced Plant Defenses, Plant Physiology, Volume 160, Issue 3, November 2012, Pages 1468—1478
- Poretsky, Elly et al. “Comparative analyses of responses to exogenous and endogenous antiherbivore elicitors enable a forward genetics approach to identify maize gene candidates mediating sensitivity to herbivore-associatedcmolecular patterns.” The Plant journal : for cell and molecular biology, 10.1111/tpj.15510. 26 Sep. 2021
- Steinbrenner, Adam D., et al. “A receptor-like protein mediates plant immune responses to herbivore-associated molecular patterns.” Proceedings of the National Academy of Sciences 117.49 (2020): 31510-31518.
- Schmelz, Eric A. “Impacts of insect oral secretions on defoliation-induced plant defense.” Current Opinion in Insect Science 9 (2015): 7-15.
Written by Kevin Martinez
Kevin is a Biochemistry and Cell Biology Major from Roger Revelle College. He will be graduating in 2022.
