By Christina Cui | UTS Staff Writer | SQ Online (2014-15)
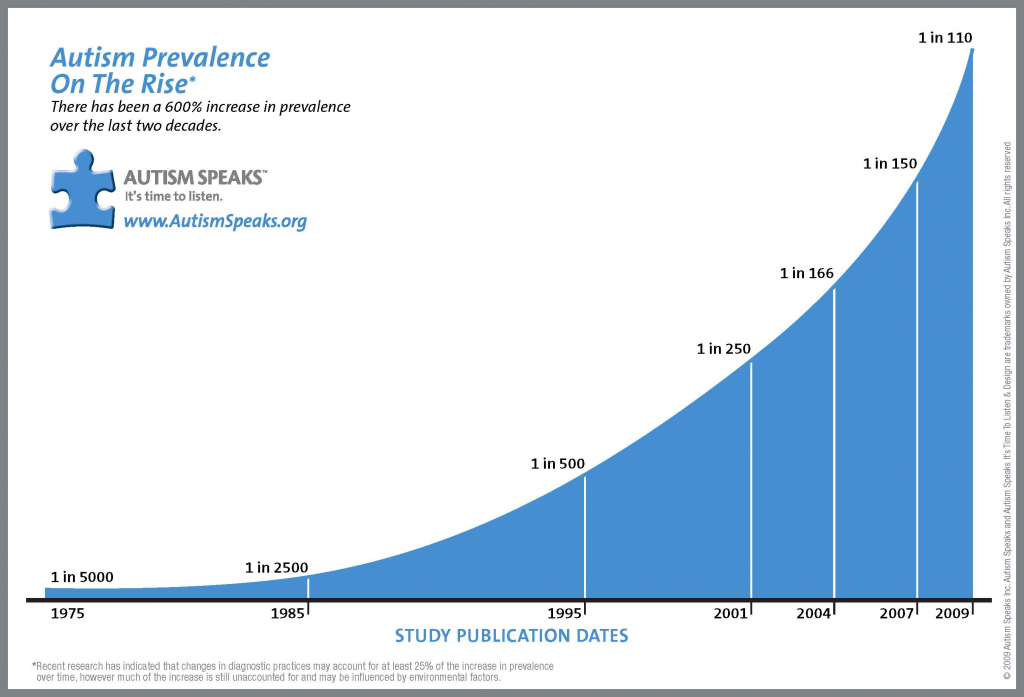
Out of an elementary class of 30 students, 5 students will have a developmental disorder. Just a couple of decades ago, autism was an obscure diagnosis handed to very few patients but, today, 1 in 68 children with be diagnosed with autism spectrum disorder (ASD)[1].
ASD’s run the gamut of developmental phenotypes, from slight speech impediments to severe difficulties in social behavior. This diversity is compounded by variable combinations of genetic and environmental factors that somehow create these different phenotypes. Clearly, each ASD case is unique; thus studying the mechanisms for ASD has been extremely difficult. That is, until the Tooth Fairy came to help. Through the Tooth Fairy Project, parents can exchange their children’s baby teeth for something more valuable than just a crisp dollar bill [2].
And in this project, the Tooth Fairy is not quite who kids might expect it to be. UC San Diego School of Medicine physician and professor Dr. Alysson Moutri collects the teeth that parents send in, not to build a tooth fairy castle but to construct models for autism. The cells in each tooth can be turned around in their development and directed to create what are known as induced pluripotent stem cells. Induced pluripotent cells (iPSCs) are essentially adult cells returned to their embryonic state so that they can differentiate into many different types of cells besides their original identity [3]. [Figure 2] Dr. Moutri redirects dental pulp cells to form neurons and, since each iPSC model contains the genome unique to an individual, these models are able to better demonstrate the diversity of ASD [4]. (Here is more information about the Tooth Fairy Project.)
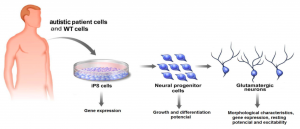
While this model has demonstrated promise in more unique models, there are still small details being ironed out in its methods. For example, the cells that are created are not guaranteed to end up in neuronal form. Some become differentiated into glia, neighboring cells in the brain that service neurons, while others remain in the progenitor, or early stage, form. But, while the model is not entirely perfect, this “autism in a dish” is a giant leap away from simple mouse models or other organisms, especially since a large component of ASD is social. Through this, researchers have studied the morphology and wiring of neurons with developmental disorder phenotypes. Some have found smaller cell bodies, others found reduced number of neuronal spines. Although the details of these results have not been fully understood, the changes in cell structure are leaps towards better understanding the mechanism behind a diverse disorder like ASD.
And, as more of the pathophysiology is uncovered, more personalized therapy options can be developed to better streamline treatments [5]. In fact, this method of modelling neurological disorders have been so effective that it has expanded to include disorders such as Williams Syndrome, resulting from a chromosomal deletion, and Rett’s Syndrome, a disorder linked to the X chromosome [6]. A total of nine billion dollars is spent every year on treating ASD. By capturing the nuances of each case, researchers will be able to utilize those resources more efficiently through personalized medicine. These findings are worth much more than a crisp dollar bill underneath the pillow.
[hr gap=”0″]
Sources:
- blog.autismspeaks.org/2010/10/22/got-questions-answers-to-your-questions-from-the-autism-speaks’-science-staff-2/
- https://www.pediatrics.ucsd.edu/research/muotri-lab/research/Pages/default.aspx
- blog.autismspeaks.org/2010/10/22/got-questions-answers-to-your-questions-from-the-autism-speaks’-science-staff-2/
- https://bbrfoundation.org/brain-matters-discoveries/a-discarded-baby-tooth-may-unlock-causes-and-treatment-for-atypical-autism
- www.sciencedirect.com/science/article/pii/S0006322315002267
- https://health.ucsd.edu/news/releases/Pages/2014-11-11-asd-tooth-fairy-project.aspx
