As of now, the use of stem cells in medicine is quite limited. Only two stem cell treatments have been approved by the FDA to date. The first is Fedratinib for treating Myelofibrosis, a rare type of blood cancer that causes scar tissue to form on one’s bone marrow—the fatty tissues at the center of bones. The second is Glasdegib for leukemia in patients over the age of 75. Currently, several active clinical trials in the United States are studying the effectiveness of stem cell therapy for ailments like Alzheimer’s disease, Parkinson’s disease, and the replacement of diseased or damaged organs in the body. Moreover, new stem cell research is emerging in the field of reproductive and developmental biology. Scientists are looking into how stem cells can further this area of study, and they have already made two groundbreaking discoveries: stem cells can be used to create synthetic embryos and they can be used to conceive a child with parents of the same sex.
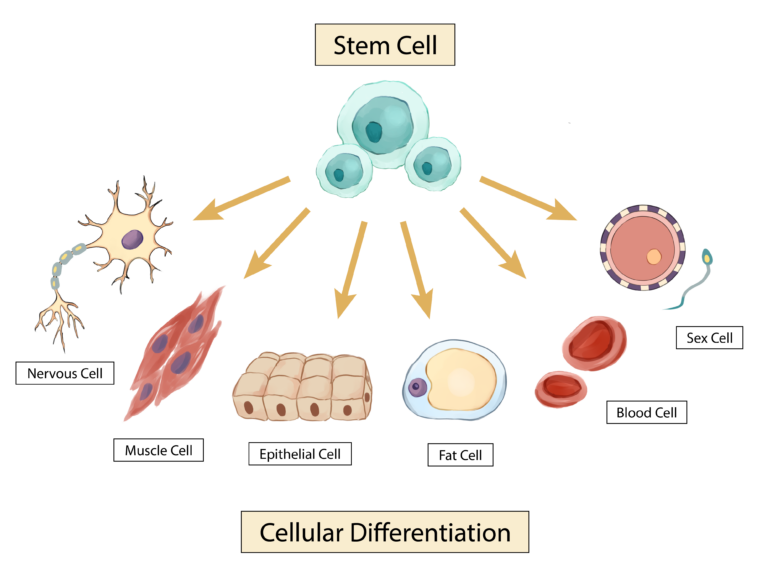
Stem cells are unique from other cells because they are unspecialized and have no set function in the body like a blood or muscle cell, but they do have the ability to develop into a variety of different types of specialized cells in the human body. There are two main types of stem cells that are currently well-understood: embryonic and adult. Embryonic stem cells are cells extracted from early embryos that are pluripotent, which means they can differentiate into any cell type in the body. Adult stem cells come in two forms, the first being from tissues in the body, like skin or brain tissue, and will only differentiate into specialized cells from the tissue in which they originate. For example, stem cells from skin tissue will turn into more skin cells, not other types of cells such as neurons. The other type of adult stem cells are known as induced pluripotent stem cells—the term ‘induced’ stems from the fact that they have been modified in a laboratory to act more like embryonic stem cells. Induced pluripotent stem cells can differentiate into more than one type of cell in the human body.
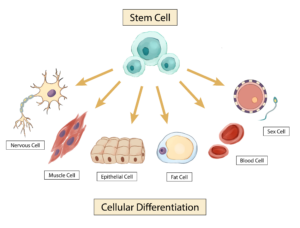
Image: Visual representation of stem cells transforming into specialized cells
Illustration by Jenny To
The Future of Developmental Biology: Synthetic Embryos
In a study done at the California Institute of Technology, Magdalena Zernicka-Goetz, a Polish developmental biologist, and her colleagues were able to use the stem cells of mice to create synthetic embryos in a manner similar to how bacteria are cultured. First, they placed mouse embryonic stem cells and extraembryonic stem cells (found outside the embryo) into well plates. These are similar to the Petri dishes that bacteria tend to be cultured in because they hold the necessary media for cell or bacterial growth and formation. While an agar medium promotes bacterial growth, several different media including FC, IVC1, DRH, and EUCM—all of which provide nutrients and other growth factors that aid in both embryo and stem cell culture—were used to promote the aggregation of the mouse stem cells into the desired synthetic embryos. In addition to requiring specific media, the cells required specific oxygen conditions (21% oxygen provided continuously or slowly increased from 5-21% over the incubation period) to further aid in embryo development, just like how bacteria are incubated at specific temperatures to help colonies grow. The fully-formed synthetic embryo models exactly replicated natural mice embryos up to 8.5 days after fertilization, and they contained both brain and heart structures. Jacob Hanna, a professor of stem cell biology at the Weizmann Institute of Science, conducted a similar study: instead of using well plates, Hanna and his team developed an electronically controlled embryo culture platform that supports stem cell assembly into embryo structures containing hearts, brains, neural tubes, guts, and germ cells. This electronic machine is made up of similar plates used in the previously described study; however, it can rotate and has customized gas and pressure regulation modules to better control the incubation period.
The successful results of Hanna and Zernicka-Goetz’s studies have enormous implications for the future of the reproductive and developmental biology fields. First, these synthetic models could replace the use of lab animals in studying early developmental stages and concepts of early development like infertility and pregnancy loss. However, there are still several limitations to this research: for example, Zernicka-Goetz’s synthetic embryoids—a structure that resembles an early-stage embryo created in a lab—are unable to survive to term, which is 20 days for mice. Hanna’s study had a similar issue, indicating that improvements need to be made to these two cell culturing methods. Getting these embryos to term would be vital for studying the later stages of development. Furthermore, there are several ethical and societal implications that must be explored and discussed before creating artificial human embryos. For instance, a current controversial debate in the bioethics community is whether or not research on synthetic embryos should be allowed past the 14-day limit set for actual embryos. This begs the question of whether or not artificial embryos should be treated as ‘life,’ significantly if they could eventually develop into living persons, like actual embryos.
The Future of Fertility: Same-Sex Conception
In addition to the development of synthetic embryos, stem cells have also aided in the creation of mice pups from two male mice. Katsuhiko Hayashi of Kyushu University and Osaka University in Japan accomplished this by converting the adult skin cells of male mice into induced pluripotent stem cells. These were then transformed into female egg cells through treatment with a drug named reversine. The eggs were then fertilized and transplanted into the embryos of female mice. 7 of the 630 embryos survived and developed into mice pups that could live on and reproduce as normal. While this is a low success rate, this is only the first study of its kind. More research is needed to truly determine if this is a viable method of conception. If it is, it could have many positive consequences. For example, it may be possible to reproduce endangered species—like the orangutan, the Asian elephant, the blue whale, the chimpanzee, the red panda, and the sea turtle—from a single male in the future.
In addition, stem cell embryos could increase the alternative fertility and reproductive options for couples who cannot naturally conceive, while potentially eliminating the negative ethical challenges of donor eggs: the principle of justice and respect for autonomy. The principle of justice is a medical ethics term that refers to the fair and equitable distribution of medical access and opportunities; in this case, donor eggs. Currently, donor eggs are more likely to go to those who can pay more for them, not necessarily those who are more fit to be parents. Likewise, respect for autonomy is another phrase commonly used in discussions of medical ethics to describe an individual’s right to make informed decisions about their own body. As of now, egg donation is highly unregulated, and donation agencies often do not properly explain the emotional, physical, and psychological implications and risks of the process to both donors and recipients. Additionally, their websites often contain predatory language to manipulate those in financial need to donate their eggs or those desperate for children to start the recipient process. For instance, a study done by Lindsay Gezinski, an associate professor of social work at the University of Utah, found that donation sites use highly emotionally-charged language compared to neutral medical terminology to describe the process of egg donation (e.g. 9 of the 19 sites studied used the phrase “the gift of life,” which is non-scientific diction). The characteristic messaging of these websites tends to neglect considerations for health complications that may occur through egg donation. One somewhat common complication is the development of Ovarian hyperstimulation syndrome—a condition characterized by the swelling and leaking of ovaries. Although most cases of ovarian hyperstimulation are mild and treatable (i.e. abdominal pain that will resolve itself within a week or two), some cases can be critical with symptoms like difficulty breathing and kidney failure.
Even though there is still much research to be done, it seems as though stem cells are the key to many future medical advancements. Not only could they be effective in treating different diseases or cancers and replacing damaged tissues in the body, but they could also aid in the betterment of both developmental and reproductive biology. The decreased use of lab animals in research, the potential for a better understanding of issues regarding infertility and pregnancy loss, and the ability for same-sex child conception are all incredible advances that would improve both science and quality of life. The studies conducted by Zernicka-Goetz, Hanna, and Hayashi are just the beginning, and much more research regarding stem cells in these fields should emerge within the next few years. Although yet to be fully realized, the nature of stem cells holds great promise for the future.
Sources:
- https://www.cirm.ca.gov/patients/power-stem-cells/
- https://my.clevelandclinic.org/health/diseases/15672-myelofibrosis
- https://my.clevelandclinic.org/health/body/22818-bone-marrow
- https://medlineplus.gov/stemcells.html
- https://www.stanfordchildrens.org/en/topic/default?id=what-are-stem-cells-160-38
- https://www.nature.com/articles/s41586-022-05246-3
- https://www.cell.com/cell/fulltext/S0092-8674%2822%2900981-3
- https://www.nature.com/articles/s41587-022-01479-9#
- https://www.bbc.com/news/health-65914934
- https://www.theguardian.com/science/2023/jun/14/synthetic-human-embryos-created-in-groundbreaking-advance
- https://www.nature.com/articles/s41586-023-05834-x
- https://phys.org/news/2023-03-revolutionary-scientists-mice-fathers.html
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5769183/
- https://drexel.edu/medicine/academics/womens-health-and-leadership/womens-health-education-program/whep-scholars/esieza-braimah/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4663984/#R14
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10310641/
- https://my.clevelandclinic.org/health/diseases/17972-ovarian-hyperstimulation-syndrome-ohss